Illumina, Secret Giant Of DNA Sequencing, Is Bringing Its Tech To The Masses
It would raise the stakes even higher, but that didn’t stop Jay Flatley, Illumina‘s then chief executive, from sending in vials of his own saliva and blood to the company’s newly opened lab. It was January 2009, and Illumina’s scientists were about to embark on their first mission to sequence a whole human genome.
The project, internally dubbed “Jaynome,” required a prescription from Flatley’s doctor in case the scientists discovered something medically relevant. It took several weeks and cost tens of thousands of dollars for the lab to deduce the sequence of the genome. “We felt the pressure, but we were doing a lot of firsts at that time,” recalled Brad Sickler, the company’s then recently hired bioinformatician. “Jay was in the first 10 people in the world sequenced at this depth.”
The result of the Jaynome experiment hinted at the potential—and some of the challenges—of bringing gene sequencing into clinical care. Flatley learned he had a condition called malignant hyperthermia, which can result in sudden death while under general anesthesia. “It’s 100 percent preventable if you know about it,” Flatley later told me. “Only few people do.” He was also informed that he shouldn’t have survived childhood. At that time, it was no easy feat to shift through a giant pool of 3.5 to 4 million variants, without having robust data to compare it to.
All told, the Jaynome project set Flatley on a path of wanting to democratize genetic information by making the tools exponentially cheaper and easier to use. And his company has played an instrumental role in doing just that.
If you’ve ever used 23andMe, Ancestry.com, or any other genetics-testing service, chances are that your genes were sequenced on machines made by the $25 billion biotech behemoth. Now the undisputed leader in the emerging field of DNA sequencing in the U.S., Illumina has outstripped its rivals by selling its sequencing hardware to medical researchers around the world.
It has been a key player in driving down the cost of genetic sequencing from $100 million in 2001 to $1,000 today. As Nature once put it, “That does not just outpace Moore’s law—it makes the once-powerful predictor of unbridled progress look downright sedate.” The process used to take weeks, but can now be finished in days—16 people’s genomes can be sequenced in just three days using one of Illumina’s sequencers, the HiSeq X Ten. That machine is capable of sequencing 18,000 human genomes each year.
The technology is quickly moving from the lab and into hospitals, clinics, and even homes with the advent of consumer genomics. And Illumina, founded in 1998, is looking for new ways to capitalize on the growing clinical and consumer markets with new applications, and become an essential part of every aspect of the DNA ecosystem.
The challenge for Illumina, as with any other large company, is to continue to innovate while maintaining a grip on its core business. The company recently took a page from Alphabet by announcing a series of “moonshot” ventures, called Helix and Grail, which will create an App Store model for DNA informatics and bring early cancer screening tests into every doctor’s office. And veteran Apple executive Phil Schiller joined Illumina’s board this summer, giving the biotech company an injection of consumer marketing know-how.
“We saw this cycle with computer tech. It used to be mainframes, then office equipment and now every phone, TV, car, and music player. They are all computers,” says Schiller, who was a systems analyst at Massachusetts General Hospital long before he took charge of Apple’s App Store. “I think that the whole category of gene sequencing is going through the potential of a similar evolution, where it moves from being in very few labs to natural business environments to medical institutions, hospitals, and eventually being the tech that we all use in our lives.”
In another big change, Flatley also stepped down this summer after 17 years at the helm. His replacement, soft-spoken MIT-trained computer scientist Francis deSouza, was recruited from Symantec to expand the company’s global footprint and build out software products to sit on top of the instruments and other services it sells. DeSouza is also spearheading some cultural changes, including offering employee benefits such as 16 hours of paid volunteer time and free access to a screening test for undiagnosed diseases.

But critics say it will be a challenge for lllumina to figure out how to expand into these same markets without encroaching on its customers’ territory. Startups have already entered the clinical market with applications for everything from “liquid biopsy” tests to monitor late-stage cancers (an estimated $1 billion market by 2020, according to the business consulting firm Research and Markets), to non-invasive pregnancy screenings for genetic disorders like Down Syndrome ($2.4 billion by the end of 2022). The majority of companies in both the clinical and consumer arenas rely on Illumina’s machines for their underlying analysis.
“I think entrepreneurs, investors, and companies are at times apprehensive about Illumina and worried about if, and when, they might choose to compete against them,” says John Stuelpnagel, Illumina’s cofounder and former chief operating officer. If it can walk that tightrope, many expect that Illumina will tap into a potential goldmine in the next five years.
“Less than 3% of the population knows anything about their genomic data,” adds Jonathan Groberg, a biotech analyst at UBS. “This opportunity is where the internet was in the 1970s.”
The Hardware In Its DNA
Illumina’s San Diego headquarters bears some resemblance to the technology campuses of Silicon Valley. The hallways are filled with light, even on the foggy day I visited, with ample space for employees to collaborate, coffee shops, and an onsite cafeteria (but the meals aren’t free, in part to demonstrate that resources are being spent wisely). The senior executives work in cubicles with everyone else, and there are no offices. In his first weeks as CEO, DeSouza reduced the size of the cubes on the executive floor even further to make it easier to collaborate. Those at the top have also blocked out time on their calendars for anyone to ask questions or suggest ideas.

Flatley, Illumina’s former CEO, is credited by current and former employees I spoke to with maintaining a “culture of innovation,” especially as the company grew to some 5,000 employees and annual revenues of more than $2 billion. But back in 2003, just five years after it was founded, the company was barely eking out a profit and facing off against some larger, tougher competition in the form of larger and better-funded companies.
After Flatley made the strategic decision to buy Solexa, a British biotech startup with a novel approach called “sequencing by synthesis,” that all started to change. From 2007, Illumina’s researchers improved on the core technology, and the company built a global distribution network. The executive team revised some of its processes to allow for rapid new product development at multiple geographic sites.
“We developed this incredible ability to innovate when our talent, processes, and product-development strategy came together,” recalls Stuelpnagel. “It was a culture of innovation fueled in part by a healthy paranoia of others trying to outcompete us.” Within five years, many onetime competitors like Roche (at one point, also a potential buyer) and Complete Genomics fell away or got acquired.
It was Flatley’s idea, in 2009, to get an edge by setting up a federally certified lab on the premises to test new hardware, and showcase what could be possible with next-generation sequencing, and even assist in the occasional medical case.
To open up whole genome sequencing to others beyond himself, Flatley initiated a consumer genomics push with the Understand Your Genome Project (UYG), which provides far deeper sequencing than a 23andMe or Ancestry (in this case, a doctor’s note is required). For under $3,000, participants have learned they have a resistance to a certain drug, like Warfarin, or a gene mutation that indicates a likelihood of getting breast or ovarian cancer. Illumina’s Erica Ramos, a full-time genetic counselor who works with UYG, told me they find out about a significant variation with about 2% of the population. “A lot of what we’re trying to do is drive up awareness of genomics in practice,” she says.
The lab, where DNA sequencing is performed internally, is filled with rows of humming computers of varying sizes. The HiSeq X Ten, which is as large as an office photocopier, sells as a set of instruments for more than $10 million. There are 35 customers globally that own one of these machines, including the Garvan Institute of Medical Research in Sydney and the Broad Institute in Cambridge, Massachusetts. They’re sometimes referred to as the “X Ten club.”
Lower-end instruments for more targeted applications, which can be as small as a desktop computer, sell to molecular biologists, pathologists, and oncology researchers for around $50,000 each.
Illumina has captured more than 70% of the sequencing market with these machines that it sells to academics, pharmaceutical companies, biotech companies, and more. During his tenure as CEO, Flatley built a reputation around tight product execution, culminating in Illumina being the first to announce the $1,000 genome in 2014.
“Nowadays, Illumina is like the 800-pound gorilla in genomics,” said Joyce Tung, 23andMe’s director of research, when asked about the company’s impact.
“I have never encountered any other machines but Illuminas,” adds Gabriel Otte, cofounder and CEO of Andreessen-Horowitz-backed genomics startup Freenome. “And my PhD is in genomics.”
Some biotech experts suspect that the core technology will eventually be commoditized as the price plateaus, and perhaps comes down further, in the coming years. But Flatley is convinced that this is far from imminent. “It will be a long time before that happens.”
These days, Illumina faces its stiffest competition from Thermo Fisher Scientific, the Waltham, Massachusetts, multinational that supplies biotech for everything from anti-doping analysis to genetically modified crop production. And Illumina may soon face off with European vendor QIAGEN as it moves into the clinical market. There are also some up-and-coming rivals in more niche areas, such as the makers of “long-read” sequencing technologies that are useful for things like looking at structural issues with chromosomes that lead to diseases like cancer, and testing for some hereditary diseases that are missed with short-read approaches.
One potential rival is U.K.-based startup Oxford Nanopore, which makes small, portable sequencers that it claims can deliver real-time reports and be used in the field, including at the International Space Station. Oxford Nanopore’s technology reads long strands of DNA by measuring changes in electrical current as a nucleotide passes through a microscopic pore. Critics of this technology point to the lack of accuracy, although it shows promise in applications that involve rapid diagnoses of diseases like the Zika virus.
It’s unlikely that Illumina will be knocked off its pedestal by Oxford Nanopore or any other startup in the near future, according to a half dozen experts I spoke to. That’s in part because Illumina has invested big in R&D, and some biotech startups like 10X Genomics are building applications to enhance the information that can be gleaned via Illumina’s sequencers. The other reason is that Illumina, like many of its former competitors, has shown that it is comfortable using litigation to remain competitive.
After investing over $18 million in Oxford Nanopore in 2009 and an undisclosed sum in 2010, Illumina divested and filed suit against the company earlier this year, claiming that the British startup’s technology contained ideas stolen from its patent. Oxford Nanopore responded by sending a letter to the U.S. International Trade Commission claiming that the the suit was analogous to “restricting the trade on smartphones because mainframe and desktop computers are available,” and that Illumina was seeking to “stifle emerging competition and enhance its monopoly.” The company also told the commission that Flatley had previously participated as an adviser on its board. A few outside observers speculated that the real battle was about whether Oxford Nanopore technology would someday displace Illumina in the battle to provide hardware and services in the sequencing market. The lawsuit was settled in August.
Others say this kind of litigiousness is par for the course in the life sciences world. Illumina, for instance, was sued by many of its larger rivals in the years that it was the underdog. Large biotech companies are under pressure from stakeholders to expand their businesses and avoid competition from startups snapping on their heels.
In its current position, Illumina is either an ally or frenemy to virtually every genomics company that exists. Even those I spoke to that harbored some fears that Illumina would encroach on its territory were reluctant to speak publicly about it.
“As Illumina moves into the clinical markets, it’s making for some tough conversations,” says Groberg.
One of Illumina’s mantras, which I heard repeatedly while reporting this story, is that it does not compete with its customers. But when the topic of its new ventures in reproductive health comes up, that concept seems to break down.
In 2013, Illumina bought a company called Verinata Health for $350 million. That sent shockwaves through the industry (one executive described the move as “puzzling”), as Verinata is a direct competitor to several of Illumina’s customers in a space known as noninvasive prenatal testing (NIPT). It’s a technique for analyzing fetal DNA circulating in the mother’s blood for signs of chromosomal conditions. The market is now valued at $1 billion, and almost a million of these tests are sold annually.
I asked Illumina’s chief commercial officer Christian Henry whether the buy-up of Verinata was intended as an explicitly competitive move against other NIPT vendors, several of whom like Natera rank among Illumina’s biggest customers. “It’s a foundational question for us how much we compete with our customers,” he responded, carefully.
Henry stressed that the company is trying to “walk the fine line” by avoiding selling directly to doctors. Instead, Illumina is choosing to sell to labs or other NIPT companies that want to use the equipment. Still, it’s easy to see why other NIPT players would be threatened, especially given the fact that the field is already so crowded and litigious. “Illumina says it wants to democratize the ability for any lab to offer these NIPT tests, but you can follow the trail as well as anyone,” adds Groberg.

The Holy Grail: Cradle To Grave Sequencing
“Today, where genomics is considered important and valuable and should be paid for is in the range of genetic and rare diseases,” says Euan Ashley, an associate professor of medicine and genetics at Stanford University and a rare disease specialist. “I think you’d find very little argument inside or outside Illumina in relating to that as a consensus position.”
Illumina is making an impact in these areas by selling sequencing tools to researchers, and occasionally clinicians who study and treat disease. But it’s a little-known fact that scientists at Illumina do take on the occasional compelling rare disease case, and it’s typically a child with a set of debilitating symptoms, where no diagnosis has been found through conventional methods.
Over coffee at the company’s cafeteria, Illumina’s senior director of scientific research, Ryan Taft, shared the story of a former patient who had a mysterious disorder of the central nervous system. As a toddler, a young Australian boy named Massimo lost the ability to eat and crawl. The boy’s father called Illumina after surmising that the disease was likely genetic.
Massimo became the 45th person in the world to have his genome sequenced by Illumina’s machines, followed by his parents. The rationale seemed simple enough: His parents were healthy and Massimo was not, which meant that some quirk in the DNA would be able to uncover the problem. But as Taft explained, he had to sequence the families’ genomes 30 or 40 times to ensure there wasn’t an error. That’s about 120 billion letters each.
The hope is that DNA sequencing might find a potential clue by uncovering the right variant(s) among a massive pool of potential culprits. “That’s like finding a needle in a big pile of needles,” Ashley says.
Taft, who is also a computer scientist, whipped up some algorithms using existing open-source frameworks (these are the kinds of sample-to-answer informatics tools that Illumina is building for clinicians). He found a mutation in Massimo’s genome that he thought might explain the symptoms. To test the theory, Taft set out to find other children that resembled Masimo and sequence them. By investigating MRI biobanks, Taft and Massimo’s family were able to uncover nine other children in the U.S. and Europe with similar symptoms. They eventually diagnosed a new disease known as HBSL.
Taft now leads the company’s internal efforts to support clinical trials and collaborate with rare disease researchers on the occasional case. “It’s a small effort, but we want to engage and try to solve these medical cases,” he says.
Because of genomics, Ashley and other rare disease specialists expect that the library of diseases with a genetic basis will drastically expand. “Different diseases will get different treatment based on their specific mutations; drugs will have more impact and trials will be more successful,” says Ashley, who is the principal investigator of the National Institutes of Health’s $7.2 million-funded Undiagnosed Diseases Network.
The hard part is making sense of all the data that will be coming off the sequencers and parsing out what’s clinically meaningful in all the noise.
For Illumina, it’s not an easy feat to jump up the technology stack from hardware to software and services. As Serge Saxonov, CEO of 10x Genomics and a cofounder of 23andMe explains, “Companies have DNA based on how they got started.” And software requires different skill sets and a new way of thinking.
Nevertheless, computing hardware—whether it’s your laptop or Illumina’s sequencers—becomes far more valuable when it’s sold as a package with software and tools that make it useful. Illumina will need to amp up its offerings to tap into the much-larger clinical and at-home market, and the company’s new CEO is under pressure to do just that.
At a recent UBS Summit in Park City, Utah, deSouza spelled out his plan to incorporate software into the sequencing service, rather than to try to sell it separately. Over lunch, deSouza shared that he had been hired in part for his expertise in developing secure software products.
One such venture currently in development is called BaseSpace, a cloud-based offering with software tools for interpreting DNA data from the company’s machines.
The company also invested in a $100 million-funded software spinout dubbed Helix, which is led by former MapMyFitness cofounder Robin Thurston. The idea behind Helix is to partner with well-known brands to incorporate genomics into targeted, personalized apps in a model that borrows from the Apple App Store. A customer might check how quickly they metabolize caffeine via an app developed by a nutrition company. Helix will sequence the customers’ genomic data and store it centrally, but the nutrition company delivers the report back to the user.
“The brands will do the heavy lifting from a marketing standpoint,” Flatley explains. For that reason, Helix’s team expects that the number of people who have been sequenced will drastically increase. Thurston predicts that it will be 90% of people within 20 years.
Another distinguishing factor is that the genome data isn’t delivered back to the customer all at once; instead, answers are delivered for specific questions as new apps are developed by Helix’s brand partners.
The model depends on Helix building a very large number of partners, so customers will want to find out insights about their genome over and over again. Helix subsidizes the cost of sequencing the whole exome for the first time, expecting that customers will query it repeatedly throughout their lifetime. Another difference to consumer genomics apps like 23andMe is that Helix offers exome sequencing, which offers more information and is more expensive than traditional “genotyping” methods.
“This will be the first deep sequencing test broadly available on consumer market,” says Thurston.
“We find the idea of Helix intriguing and are interested to see how companies use their platform,” says a spokesman from 23andMe, when asked whether 23andMe views Helix as a competitor at this stage. “It also validates our confidence in the consumer genetics market that we’ve pioneered for a decade.”
Helix is expected to launch with its first set of customers, which include Mayo Clinic, LabCorp, and Duke University later this year.

“I’m Confident This Is The Future”
In 2015, the White House unveiled its $215 million-funded “Precision Medicine Initiative,” which is primarily focused on cancer.
The basic premise is that each tumor harbors a different set of mutations, and new therapies can be designed based on which mutations are present, with each patient receiving a personalized treatment plan by their physician. “The cancer world has fundamentally changed,” says Maurie Markman, a physician, ovarian cancer researcher, and the president of medicine and science at Cancer Treatment Centers of America. “I’ve been doing this for over 30 years and I’m a very conservative person, but I’m confident that this is the future.”
Illumina seems to agree: Its other $100 million spinout venture is developing the first FDA-approved screening test for cancer. Grail, a standalone company, will proactively test patients for the earliest signs of cancer, so they can be treated before the disease advances. The idea is to take a high-resolution snapshot of patients’ genetic fragments from dying cells of tumors that circulate in the blood. Such tests already exist to monitor patients already known to have cancer, but not to diagnose it at a much earlier stage.
That premise helped Illumina recruit Jeff Huber from Google as CEO of Grail. Huber lost his wife to advanced colon cancer in November of last year. “It was effectively a death sentence,” he says.
Flatley, who is helping to oversee the initiative while shifting into the executive chairman role at Illumina, is confident that the new company can overcome the myriad technical challenges—for one thing, the test needs to be highly accurate or clinicians risk misinforming patients. Flatley boasts that Illumina is the only company that has the technology to sequence to the required breadth and depth. “The market is a moonshot; the tech isn’t,” he says.
By 2017, Grail aims to kickstart “the largest clinical trial ever conceptualized,” says Flatley, and for the screening product to be approved to hit the market by 2019 for a price point of less than $1,000. Bill Gates and Jeff Bezos’s Bezos Expeditions have both invested in the startup, and its potential market size has been touted as $20 billion to $40 billion.
If Grail and Helix are successful, a lot more people will have access to their DNA throughout their lives. It’s no surprise that the final phase of Illumina’s masterplan has been dubbed internally as “Genomics Everywhere.” The vision is that it will be commonplace for a person’s sequencing journey to begin before birth and continue throughout their entire life.
“I think many of us, myself included, have imagined a future where genomics touches every individual in the health system,” Ashley, the Stanford geneticist, explains.
Several genomics companies are founded on this vision, particularly when it comes to leveraging technology to better treat the sickest patients with more targeted therapies. Many researchers believe that preventative care will also be more routine when patients understand (and can take steps to proactively reduce) their unique set of disease risks, or they get screened for cancer well before it spreads.
What comes next is writing DNA, and not just reading it. Gene-editing tools like CRISPR-Cas9 are making it cheaper and faster to move genes around, which has untold consequences for changing the environment and treating disease. Stuelpnagel believes that in the coming decade, Illumina may have more impact on health and agriculture than any other company. “DNA is the foundation of life, and Illumina is enabling us to learn about it and, coupled with DNA writing technologies, manipulate it.”
As the technology advances, bioethicists, consumers, and policymakers will carry a huge burden in determining how and where to draw the line.
As genomics hits the mainstream, the company knows that it needs to become a voice. This year, it hired a government affairs team and held introductory meetings with policymakers to discuss the Precision Medicine Initiative and funding for scientific research. Flatley has also stepped up his presence in Washington, D.C., especially as the company explores opportunities in the regulated clinical market. Explaining adenine, guanine, cytosine, and thymines to a broad audience without raising the ire of the FDA is no easy feat.
“I often joke that we need to do a Super Bowl ad,” says Illumina’s Henry. “Conceptually we know we need to get the message out.”
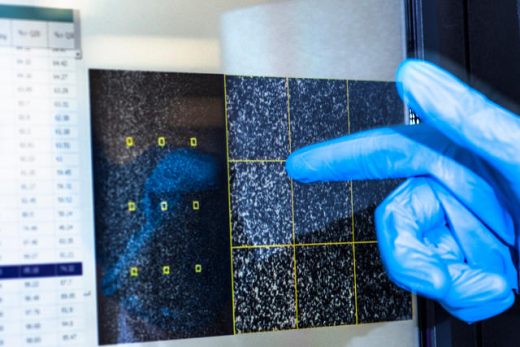
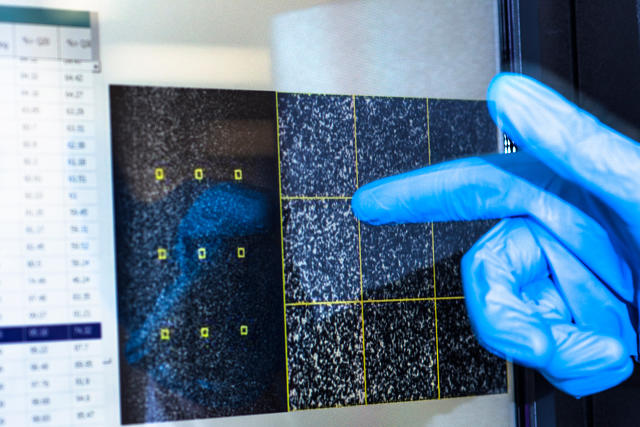
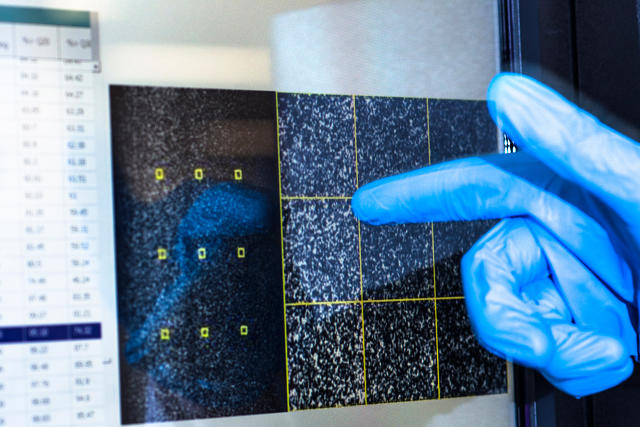
[Photos: Noel Spirandelli for Fast Company]
The first step of the sequencing process is to extract DNA from human cells, each of which contains a single copy of your genome.

From there, it is sliced into fragments less than 1,000 letters long as it is too large to be read all at once.

These fragments are attached onto a flow cell, which is like a glass slide but with chemical hooks to hold the DNA in place.

They are duplicated many thousands of times over to create colonies of the exact same sequence.

The sequence of the DNA is then read by adding fluorescent “glowing” nucleotides, individual DNA letters, that bind to a fragment.

A camera captures these reactions occurring all at once on an area about a quarter of the size of your phone screen.

Finally, the scientists will use algorithms to decode them from dots of light to individual DNA letters.

The sequenced fragments are then mapped to the human reference genome.

Illumina can now sequence 16 genomes in just three days, a process that requires immense computational power.

Its machines range in size from an office photocopier to a desktop computer.

Illumina’s internal lab team steps in to assist on the occasional medical case, often a child with a rare disease.

The company says that its machine can detect genetic variation of all types, from a single letter to millions of letters altered.

Fast Company , Read Full Story
(85)